|
6/21/2023 0 Comments Carbon dating examples 
The neutrons resulting from the cosmic ray interactions participate in the following nuclear reaction on the atoms of nitrogen molecules (N 2) in the atmospheric air: Carbon-14 has a half-life of 5730 years and would have long ago vanished from Earth were it not for the unremitting cosmic ray impacts on nitrogen in the Earth's atmosphere, which create more of the isotope. In addition, there are trace amounts of the unstable isotope carbon-14 ( 14C) on Earth. Ĭarbon has two stable, nonradioactive isotopes: carbon-12 ( 12C), and carbon-13 ( 13C). Atmospheric nuclear weapon tests almost doubled the concentration of 14C in the Northern Hemisphere. The New Zealand curve is representative for the Southern Hemisphere, the Austrian curve is representative for the Northern Hemisphere. Plants fix atmospheric carbon during photosynthesis, so the level of 14C in living plants and animals equals the level of 14C in the atmosphere.Ītmospheric 14C, New Zealand and Austria. One of the frequent uses of the technique is to date organic remains from archaeological sites. He first demonstrated the accuracy of radiocarbon dating by accurately measuring the age of wood from an ancient Egyptian royal barge whose age was known from historical documents. In 1960, he was awarded the Nobel Prize in chemistry for this work. Libby estimated that the steady state radioactivity concentration of exchangeable carbon-14 would be about 14 disintegrations per minute (dpm) per gram. The technique of radiocarbon dating was discovered by Willard Libby and his colleagues in 1949 during his tenure as a professor at the University of Chicago. Such raw ages can be calibrated to give calendar dates. uncalibrated, radiocarbon ages are usually reported in radiocarbon years " Before Present" (BP), "Present" being defined as AD 1950. Potassium or uranium isotopes which have much longer half-lives, are used to date very ancient geological events that have to be measured in millions or billions of years.Radiocarbon dating is a radiometric dating method that uses the naturally occurring radioisotope carbon-14 ( 14C) to determine the age of carbonaceous materials up to about 60,000 years. Dates derived from carbon samples can be carried back to about 50,000 years. The fact that carbon-14 has a half-life of 5,730 years helps archaeologists date artefacts. 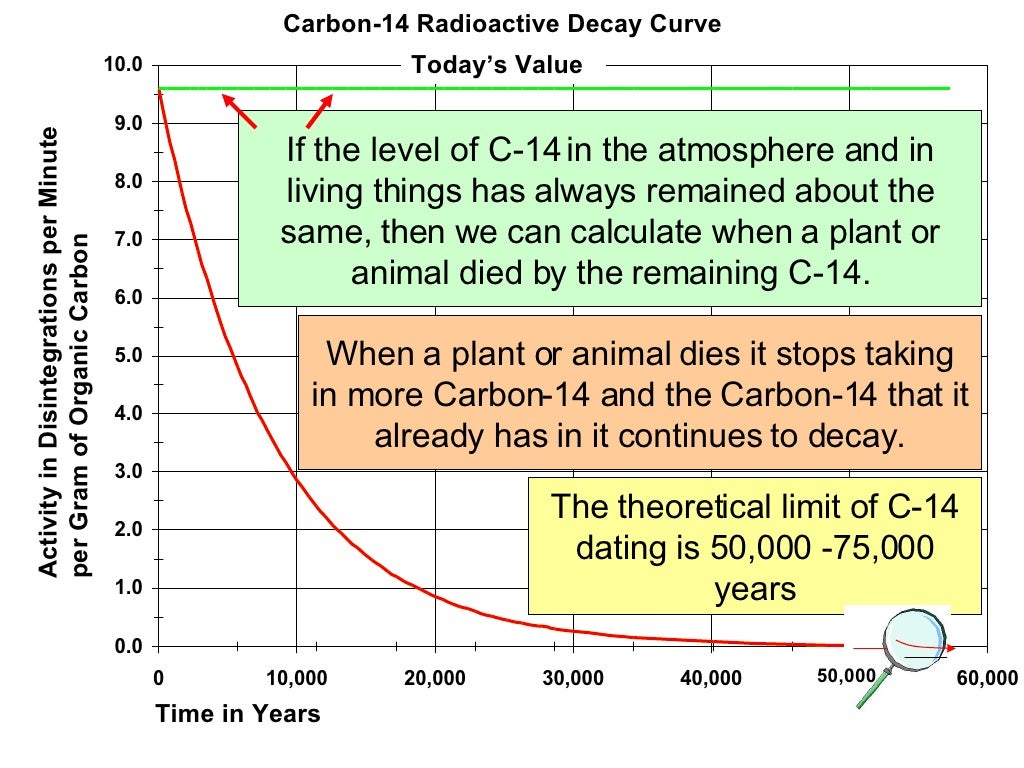
When a particular fossil was alive, it had the same amount of carbon-14 as the same living organism today. It can be deduced then that today's readings would be the same as those many years ago. The assumption is that the proportion of carbon-14 in any living organism is constant. However knowing how many carbon-14 atoms something had before it died can only be guessed at. The half-life of carbon-14 is 5,730 years. So we only have to know two things, the half-life of carbon-14 and how many carbon-14 atoms the object had before it died. In 2020, there would be 250 left, and in 2030 there would be 125 left.īy counting how many carbon-14 atoms in any object with carbon in it, we can work out how old the object is - or how long ago it died. For instance, if 1000 atoms in the year 2000 had a half-life of ten years, then in 2010 there would be 500 left. Half of the available atoms will change in a given period of time, known as the half-life. Radioactive atoms decay into stable atoms by a simple mathematical process. Carbon-14 is radioactive and it is this radioactivity which is used to measure age. Carbon-12 makes up 99% of an atom, carbon-13 makes up 1% and carbon-14 - makes up 1 part per million. They are carbon-12, carbon-13 and carbon-14. 
Different atoms of the same element are called isotopes. Certain chemical elements have more than one type of atom. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
